Hepatitis B Viruses Discovered in Ancient Human Remains
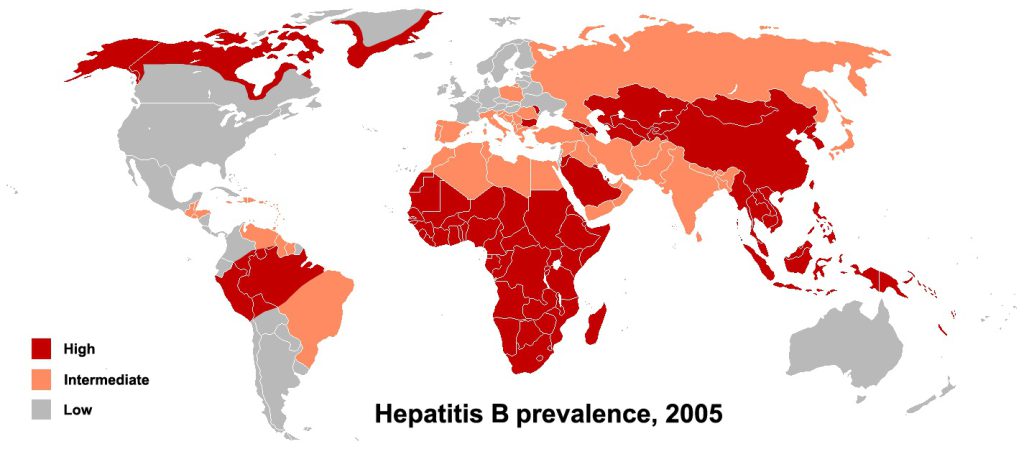
Around the world, more than 200 million people live with an infection from the hepatitis B virus, a pathogen that can reside in the human body for long stretches of time and cause serious complications, such as chronic liver disease. Despite its prevalence, little is known about the ancestral roots of the virus. New findings, published today in Nature, reveal some of the oldest samples of the virus to date—between 800 and 4,500 years old—and provide fresh insights into its origin and evolution.
Although scientists have previously uncovered the hepatitis B virus in two 16th-century mummies, most investigations into the pathogen have focused on DNA sequences extracted from humans during the last 50 years, says Barbara Mühlemann, a doctoral student studying viral evolution at the University of Cambridge in the U.K. So uncovering these ancient genomes was “a bit like finding a fossil for the first time,” Mühlemann says.
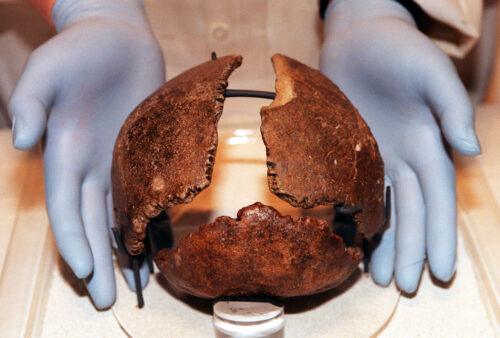
To probe for signs of ancient outbreaks in individuals who occupied Eurasia from the Bronze Age to the medieval period, the researchers examined DNA extracted from the remains of 304 ancient humans. These samples, which spanned a range of more than 6,000 years, were from large collections of skeletons from two previous examinations of human genomes in this region.
Approximately half of those ancient DNA samples came from another new Nature study, published today, that reports an analysis of the genomes of people who lived on the Eurasian steppe from the period right after the Bronze Age through medieval times. These types of studies are only possible because of the advances in genetic techniques and decades of painstaking work by archaeologists who excavated the burial mounds to unearth the remains, says Peter de Barros Damgaard, a doctoral student in molecular anthropology at the Natural History Museum of Denmark, and co-author of both of the new studies. “We were hugely privileged to work with all these skeletons at once.”
Read more from the archives: “Five Human Species You May Not Know About.”
When the researchers set out to search for viruses within these samples, they were not sure what they would discover, says Mühlemann, a co-author of the hepatitis study. While other groups had found signs of bacteria from ancient plagues in similarly aged samples, no one had looked for viruses. “We didn’t know if they would be preserved for such a long time,” she notes.
So it came as a surprise when the team found that 25 individuals showed signs of a hepatitis B infection, and that of those, only 12 of the viral genomes, which were between 800 and 4,500 years old, were intact enough for further analysis. While these are some of the oldest hepatitis viruses identified in ancient human remains to date, it is not particularly surprising that people were infected with this pathogen over thousands of years, Mühlemann says.
Further analyses of the genomes of these ancient viruses revealed that most of them could fit into a modern-day genotype, or category based on similarities in their DNA. Three of the genetic sequences, however, did not have a modern match and were linked to at least one newly uncovered strain that is now extinct. “We have really no idea of what diversity [of viruses] was in ancestral populations of humans,” says Hendrick Poinar, an evolutionary geneticist at McMaster University in Canada, who was not involved in this research. Evolutionary theory predicts that some viral sequences will be lost by random chance, and finding an extinct strain of hepatitis B suggests that the diversity of the virus may have been much greater in the past than it is today, he explains.
The researchers also found that some of the ancient viral sequences came from regions not typically associated with their specific genotypes today. For example, some scientists have proposed, based on its modern-day distribution, that the hepatitis B genotype A (a particular strain) originated in Africa and spread through the slave trade between the 16th and 19th centuries. The new findings, however, reveal that this strain of the virus existed in two approximately 4,000-year-old Eurasian samples and in one Hungarian Scythian sample that was around 2,600 years old.
“Our data suggest that the geographic origin of genotype A is not in Africa,” Mühlemann says. However, she adds, it’s still possible that the virus moved to other parts of the world through the slave trade, or that an earlier version of the pathogen in Africa remains undiscovered.
Further insights into the dynamics of ancient human groups may also help shed light on how the virus evolved. Findings from the new human genome study, for example, reveal that at least with respect to the Scythians, who occupied the Eurasian steppe from 800 to 200 B.C., previously established notions of large-scale migrations may be incorrect. Rather, the researchers’ discovery of genetically similar groups suggests that change may have occurred through smaller-scale movements. If you take these insights and apply them to the hepatitis work, it raises the question of how, exactly, these types of interactions affected the spread of ancient hepatitis infections, says Michael Frachetti, a professor of archaeology at Washington University in St. Louis, who was not involved in the work.
Mühlemann marvels that these ancient DNA sequences “open a window into studying virus evolution that we haven’t had before.” And by revealing modifications that have occurred in the past, she says, “these studies could shed light on how the virus might change its genome in the future.”