Does Your Microbiome Shape Your Friendships?

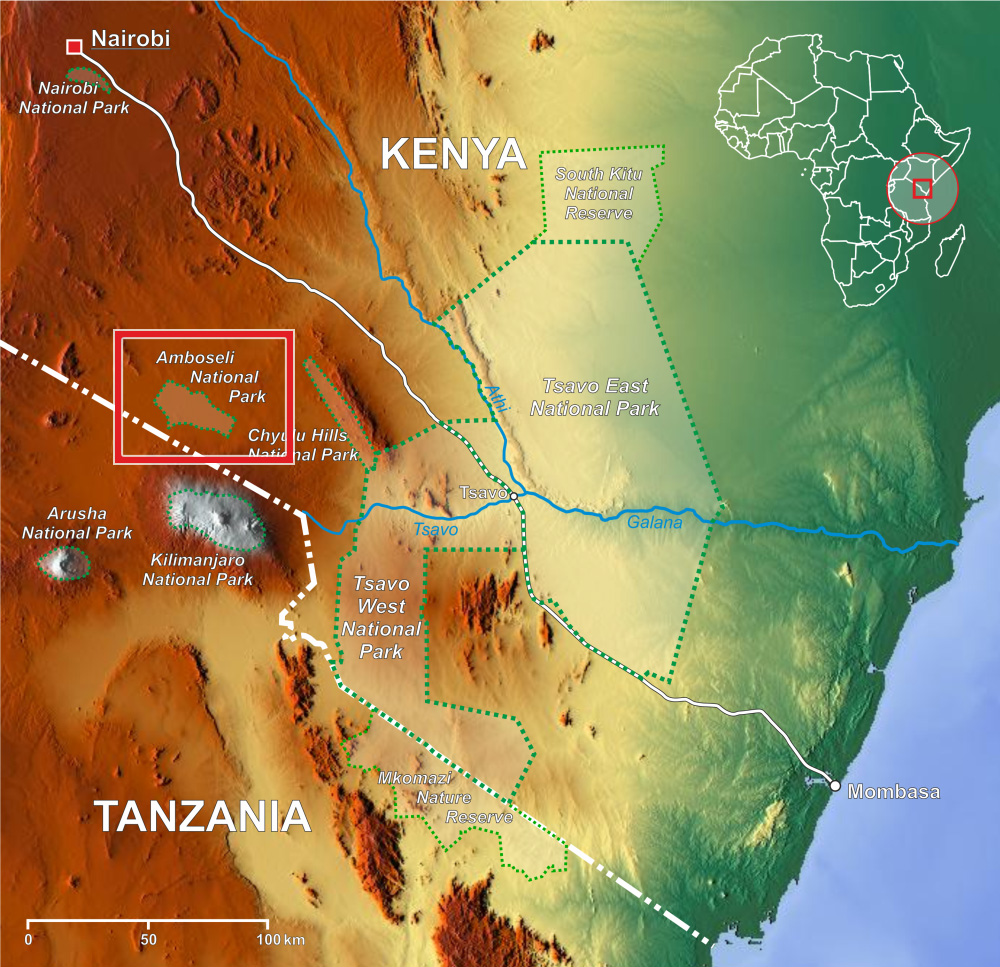
It is early morning on a wide plain in Amboseli National Park in southern Kenya. With a small Dixie cup and a wooden tongue depressor, Susan Alberts picks up a fecal sample left by a female baboon named Yoruba.
Alberts is an eminent primatologist. She is both the chair of the department of evolutionary anthropology and a member of the biology department at Duke University, and the co-director of the Amboseli Baboon Research Project. But this morning, she has the less-than-glamorous job of preparing Yoruba’s poop.
Alberts carries the cup to her makeshift field laboratory—the hood of a mud-spattered 4×4—and divides the sample among several cups, marking each with identifying details. She then treats each sample with specific chemicals according to how it will be used. “That’s for Beth,” Alberts says, as she adds some formalin to one of the Dixie cups. Beth Archie is a biologist at the University of Notre Dame and an associate director at Amboseli who heads the project’s microbiome research.
When the Amboseli Baboon Research Project was founded in 1971, microbiome research was not on the radar. The goal was to uncover the deep evolutionary roots of social behavior in primates. Since then, Amboseli scientists have followed thousands of baboons, made many important discoveries about the importance of social bonds, and been published in prestigious scientific journals such as Science and Nature. They have also picked up a lot of baboon poop. For 20 years now, the fecal samples have been used for testing DNA and steroidal hormones.
But about five years ago, those fecal samples, combined with the project’s detailed records of baboon social interactions, revealed a surprising link. Bodies host a wealth of microorganisms, which scientists call the microbiome; fecal samples specifically offer insight into the gut’s microbiome. Scientists had long assumed that the composition of the gut microbiome was largely determined by diet and environment, but the Amboseli samples revealed that a baboon’s social life is an important predictor of this microbiome.
That finding has stimulated new ideas about why animals, including humans, are social in the first place. One theory is that sharing microbes could have benefits that previously went unrecognized and that those, in turn, over evolutionary timescales, prompted us to interact with one another. In other words, our social lives shape our microbial populations—and those microbes might have helped shape our species’ social lives.
If that’s the case, every sneeze, kiss, or slap on the back could be part of a larger story, a driver of our social nature, and perhaps more beneficial than one might otherwise expect. The anthropologists and biologists who study Amboseli’s baboons are uniquely positioned to explore that idea.
Every animal—from a bumblebee to a human being—has a microbiome, or, rather, several microbiomes. The digestive system, the skin, and other parts of the body host communities of microbes that together make up the gut microbiome, the skin microbiome, and so on. In the last 10 to 15 years, advances in DNA sequencing technology have made it possible to see the surprising variety of these microbiomes with new clarity. “Everyone was astounded by what that diversity looks like,” Archie says.
There is also increasing appreciation for the good that microbes can do. While some have negative effects such as triggering disease, others are neutral or even positive. Among other things, they strengthen animals’ immune systems, produce vitamins, aid with digestion, and prevent the growth of harmful bacteria.
Until quite recently, however, research on the connection between the microbiome and sociality focused on pathogens and infection. Anyone who has ever had a child in day care or ridden on a plane with someone who is coughing and sneezing is keenly aware that microbes can transmit disease from one individual to another through physical contact or shared environments. Some researchers have suggested that fear of infection may be an explanation for humans’ deep-seated wariness of strangers.
Sharing microbes could have previously unrecognized benefits, and those, over evolutionary timescales, may have prompted us to interact with one another.
But there have been increasing signs that microbe-sharing among social partners is doing something else as well. Bumblebees, for instance, can be infected by a virulent parasite that is particularly harmful to bumblebee queens. In 2011, researchers discovered that bumblebees carry a microbe that is socially transmitted through the hive that protects the bees against this parasite—a clear case of beneficial social transmission.
And microbes may shape the way certain organisms interact. In some rodent studies, the presence or absence of specific bacteria can determine whether or not mice exhibit social deficits, such as avoiding interaction with their fellows. Gut bacteria play a part in attracting fruit flies to their mates. And microbes can influence the body’s production of hormones such as oxytocin, which plays a powerful role in bonding.
As for humans, we know that people who share a house also share microbial inhabitants. In 2014, when microbiologist Jack Gilbert, then at the University of Chicago and Argonne National Laboratory, and his colleagues studied seven families and their homes for more than six weeks, they found that the microbial communities in each home were easily distinguished from each other and that each was identifiable by family. Three families who moved during the study took their “microbial aura” with them. But Gilbert and his colleagues could not determine how much of this microbial overlap came down to shared environment, diet, genetics, or social interaction.
In Amboseli, the scientists follow hundreds of baboons on a near-daily basis, recording who does what to whom and what happens as a result. About five years ago, they began to wonder if the same social behavior that they were tracking so assiduously could tell them something about the microbiome, and vice versa.
“It was very well-established that diet played a big role in shaping the microbiome, especially in the gut, for obvious reasons,” says Jenny Tung, another associate director at Amboseli who, like Alberts, is a professor in the evolutionary anthropology department at Duke. “But what about how nice animals are to each other?” she asks. “What about their social relationships? Is that even a possibility?”
In 2015, the Amboseli baboons provided an answer. Wild baboons live in social groups of anywhere from 20 to 150 animals. Within each group, individuals vary in how social they are, but most have a few preferred animals, often relatives, with whom they groom, picking bugs and dirt out of one another’s fur. (For baboons and most other nonhuman primates, grooming is precious social currency.)
Archie, Tung, and their colleagues used a DNA sequencing technique on fecal samples from 48 baboons in two different social groups to identify the microbes found in the animals’ guts. They discovered that an individual’s social group and social network are strong indicators of which microbes will live in that individual’s gut microbiome—even when considering the roles of diet and kin. “What was surprising was how clear of a predictor it was,” Tung says.
The Amboseli baboons make a good proxy for humans for several reasons. One is that they live on the same East African savannas where humans likely evolved. “They may provide us with a snapshot that may be as close as we can get to a very early human,” Archie says.
And because the baboons’ environments are less complex than those of humans, the researchers are able to more precisely determine the way similar microbiomes reflect levels of interaction between grooming partners. In other words, they can take a lot of confounding variables that bedevil research on humans out of the equation.
The results in Amboseli were game-changing. “That was the first paper that laid it out pretty clearly that the amount of time you spend with somebody will determine how similar your microbiomes are,” biological anthropologist Katherine Amato of Northwestern University says. “And that generated a lot of excitement in terms of thinking about how microbes might be passed among individuals.”
More evidence of the links between sociality and the microbiome continued to pile up. A 2016 study in chimpanzees also showed a role for social relationships in shaping microbiomes. Andrew Moeller, an evolutionary biologist at Cornell University, found that the chimps’ social interactions increased the diversity of microbes in an animal’s gut and that each social group had a pan-microbiome, a community of microbe species that spanned the larger group.
Then in 2017, the Amboseli researchers showed that the gut microbiomes of male baboons, who change groups upon sexual maturity, become progressively similar to new group mates over a period of months. If diet rather than social relationships drove this switch, you’d expect it to happen faster, Archie says.
In a 2017 study of howler monkeys, Amato found that the more time two individuals spent near each other, the more similar their gut microbiomes were. “It wasn’t a result of kinship,” Amato says. “It was sociality.”
Now that it is clear that social behavior plays a role in shaping the gut microbiome, the next question is whether microbiomes have had a meaningful impact on our social worlds. Scientists still do not have an answer, but they are tantalized by the possibilities, which could have implications for understanding the evolution of sociality.
There are long-standing theories about why animals, including humans, should be social. Living in groups is a way to avoid predation and to get help finding food and raising offspring. Most likely all of these reasons work together to drive us toward sociality, says Archie. “There are many good and compelling reasons for why a lot of animals are social. The consequences for your microbiome might be one cost and benefit of sociality we haven’t thought of yet,” she adds.
Why would the microbiome be helpful? There are quite a few theories. It could be that social connections alter our microbiome in ways that heighten immunity. Our friends could also serve as microbial reservoirs. We pass bugs to them, and then, should we get depleted for some reason—illness, say—they can pass them back.
Having a diversity of social relationships might ensure a more diverse microbiome. And it could be that certain beneficial microbes are only obtainable from social partners.
“Should you fist bump with your friends? Should you kiss on the cheek?” Jack Gilbert asks.
Yet another possibility is that microbes play a role in social communication through scent. Humans have body odor and a lot of animals have scent-marking glands. Decades ago, researchers proposed that it is not animals themselves that make the odor but microbes, which produce volatile organic compounds that spread through the air to produce the actual smell. “If we’re using smell to communicate something about each other, it’s likely the microbes are the middle man in producing communication,” Archie says.
Of course, it’s also possible that there isn’t some larger evolutionary benefit at play in our socially influenced microbiomes. Researchers recognize the possibility that gains or losses associated with sociality and the microbiome affect animals with simpler gut microbiota, like bumblebees, but might not matter further along the evolutionary chain. “Whether that percolates up to the level of humans and other social mammals, we don’t have the evidence yet,” Tung says.
If anyone will be able to collect evidence to give these theories a firmer foundation, it’s likely to be the group at Amboseli. Around the time they published their first paper linking sociality and the microbiome, Alberts, Archie, and their colleagues realized they were sitting on a microbial gold mine.
In addition to the new samples they were continuing to collect, they had more than 20,000 freeze-dried fecal samples sitting in several freezers in the Princeton University laboratory of the Amboseli Project’s founder, Jeanne Altmann. Altmann never throws anything out, and she had continued to store what was left of the powdery poop samples after they had been used for testing DNA and hormones. This waste of the waste now turns out to be an unparalleled resource.
When Gilbert, who had studied human “microbial auras,” read the 2015 paper from Amboseli, he called Archie immediately to see if they could work together. “It’s exceptionally unusual to see something on this scale,” says Gilbert, now at the University of California, San Diego. He recalls telling Archie: “I need this, and I need it now. Give me the poop. Give me the poop.”
The decades-old samples have traveled a long way from the hoods of 4x4s in the East African savanna. After rescuing the samples from Altmann’s freezers, Archie, Tung, and their colleagues engaged in a Herculean effort to prepare them for DNA sequencing. There were so many samples they split them between Archie’s lab at Notre Dame and Tung’s lab at Duke. Every member of each lab and even spouses pitched in to carefully place the freeze-dried poop into more than 200 sample trays—each tray is about the size of an index card, with 96 wells about half a centimeter wide. Then the samples moved to Gilbert’s former lab at Argonne National Laboratory.
Archie, Tung, and Gilbert’s initial analysis served to identify the microbial diversity in each sample so that it would be possible to see how similar individual baboon’s microbiomes were. But because the samples span nearly 15 years in the lives of some animals, they can also be used to show changes in the microbiomes of individuals over time. “What we have so far is basically a couple of snapshots,” Archie says. “The hope is to turn it into a movie. How does your social life shape your microbiome as you move through time?”
The team is also doing a complete genomic analysis on a smaller subset of samples from about 400 animals. That will provide more information about the functions specific microbes have for their hosts.
If the team uncovers a link between increased microbial similarity among social partners and reproductive success, that would be evidence of an evolutionary driver at work in the connection between sociality and microbes. Gilbert hypothesizes: “If you share more bacteria within a troop, you’re going to have more offspring. That [would provide] further evidence of an evolutionary selective pressure toward that behavior and [suggest] that microbial sharing may be driving that selective pressure.”
All told, this new line of work, says Gilbert, “brings new strangeness to the phrase ‘friends with benefits.’” It also begs the question of what potential health benefits interactions with our friends might have.
“Should you fist bump with your friends? Should you chest hug? Should you kiss on the cheek? Should you hold hands?” he says. “A lot of cultures do all these things. We just don’t know. That’s the reason we do the research and try to figure that out.”